|
Carbon and oxygen form one sigma bond and two pi bonds. SO2 is an AX2E type molecule, with 2 surrounding atoms i.e oxygen, and 1 lone pair of sulfur. Here, A central atom, X surrounding atoms and E the lone pairs. We can easily find out the molecular geometry of any compound using the given chart. We will find the hybrid orbitals formed to predict the geometry of the molecule: Valance electrons of C=4.ĬO Molecular Geometry Carbon monoxide is a linear molecular geometry, there is a triple bond between C and O, and each atom contains one lone pair of electrons. The molecular geometry of SO2 is bent, with a bond angle of 120. Molar mass of sulfur dioxide 64.066 g/mol. There are 5 lone pairs of electrons in the molecule of SO2. 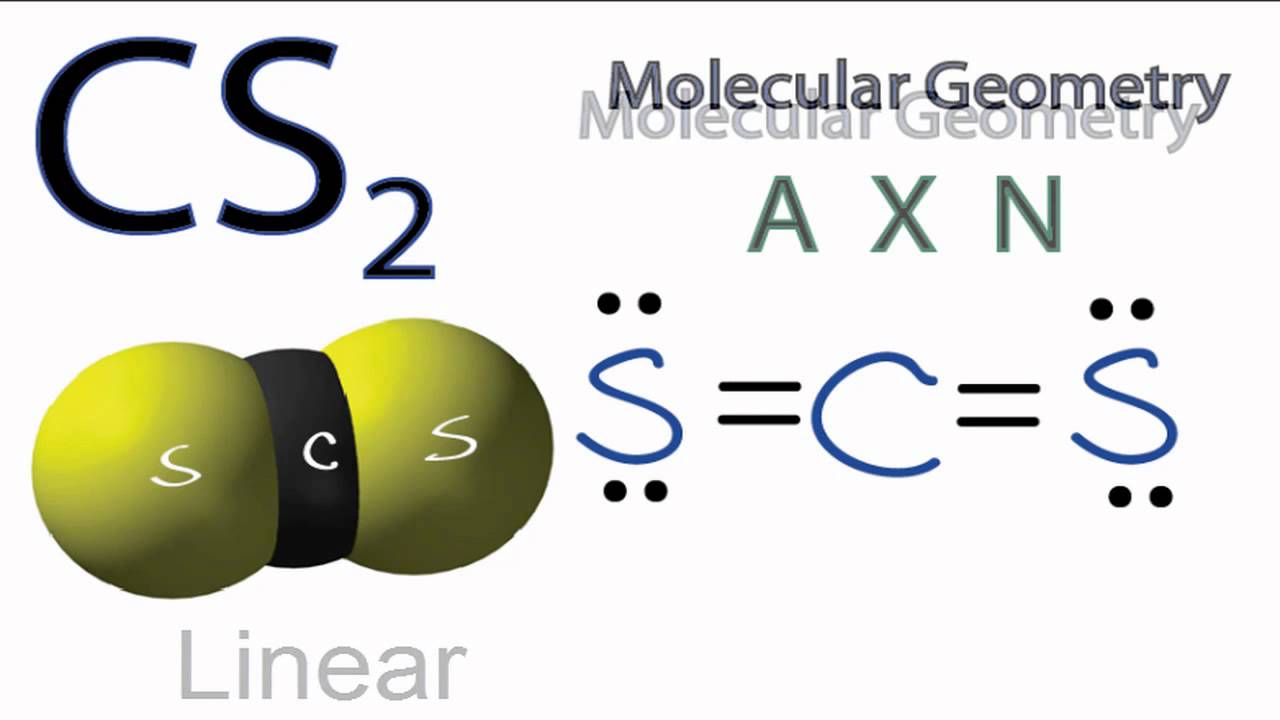
Sulfur dioxide molecule has two double bonds between the Sulfur atom and Oxygen atoms. Sulfur to the Oxygen ratio in Sulfur dioxide is 1:2. Hence, the molecular shape of CS2−3 C S 3 2 − is Trigonal Planar. The molecular geometry of sulfur dioxide is a bent shape. Hence, the molecule is sp2 s p 2 hybridized with all three bond pairs. The carbon atom is between the sulfur atoms. cs2 has 2 electron domains, resulting in a linear electron domain geometry. The electronegativity difference among the carbon and sulfur atom is nearly 0.03, and thus the bond between them is hardly even polar.ĬS2 molecule has a linear shape with bond angles of 180o. it has a linear geometry arrangement like scs. What is the molecular geometry for carbon disulfide? Is CS2 molecular or ionic?ĬS2 is a covalent molecule as both the carbon and sulfur atoms have nearly the same electronegativity values. The molecular geometry of CS2 is linear with symmetric electron region distribution around the central atom. The CS2 molecule has a linear geometry shape because it contains two sulfur atoms in. What is the molecular geometry of carbon disulfide CS2? The S-C-S bond angle is 180 degrees in the linear CS2 molecular geometry. Both electron domains are bonding pairs, so CO2 has a linear molecular geometry with a bond angle of 180°. What is the molecular geometry shape of CO2?ĬO2 has 2 electron domains, resulting in a linear electron domain geometry. The molecular geometry of CS2 is AX2 or linear. BN SiS2 CS2 SF2 N2 BF3 XeF4 6 Geometry and Polaritydoc - Molecular Geometry CO2 OF2. There are 2 atoms and no lone pair around the central atom, which corresponds to AX2 or linear. OF2 Lewis Structure, Molecular Geometry, Hybridization, Polarity. In this case, the carbon atom is surrounded by two regions of electron. In this way by determining the total electron count of the central atom you can determine the geometry of a molecule.What is the molecular geometry of CS2 enter the molecular geometry of the molecule? Now, molecular geometry is determined by the hybridization of the central atom. These bonds will account for 8 of the 16 valence electrons of the molecule. The most important part in this kind of question is to know the valence shell electron count of the central atom and the contribution of the neighboring atoms to the electron count of the central. The central carbon atom will form double bonds with the two sulphur atoms. The bond angle between oxygen and chlorine atoms (O-Cl) is 110.9° which gives the molecule a bent or V-shaped. It will use one s and one p orbitals to form the hybrids, and the remaining p-orbitals to form pi bonds with the two sulphur atoms. Through the molecular geometry diagram, one can study hybridization, polarity, and molecular orbital structure of the molecule determining the behavior of the valence electrons undergoing bond formation. This means that its steric number will be equal to Two. In this case, the carbon atom is surrounded by two regions of electron density, one for each double bond it forms with the sulphur atoms. 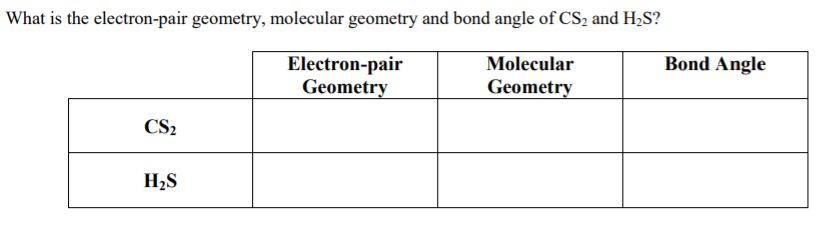 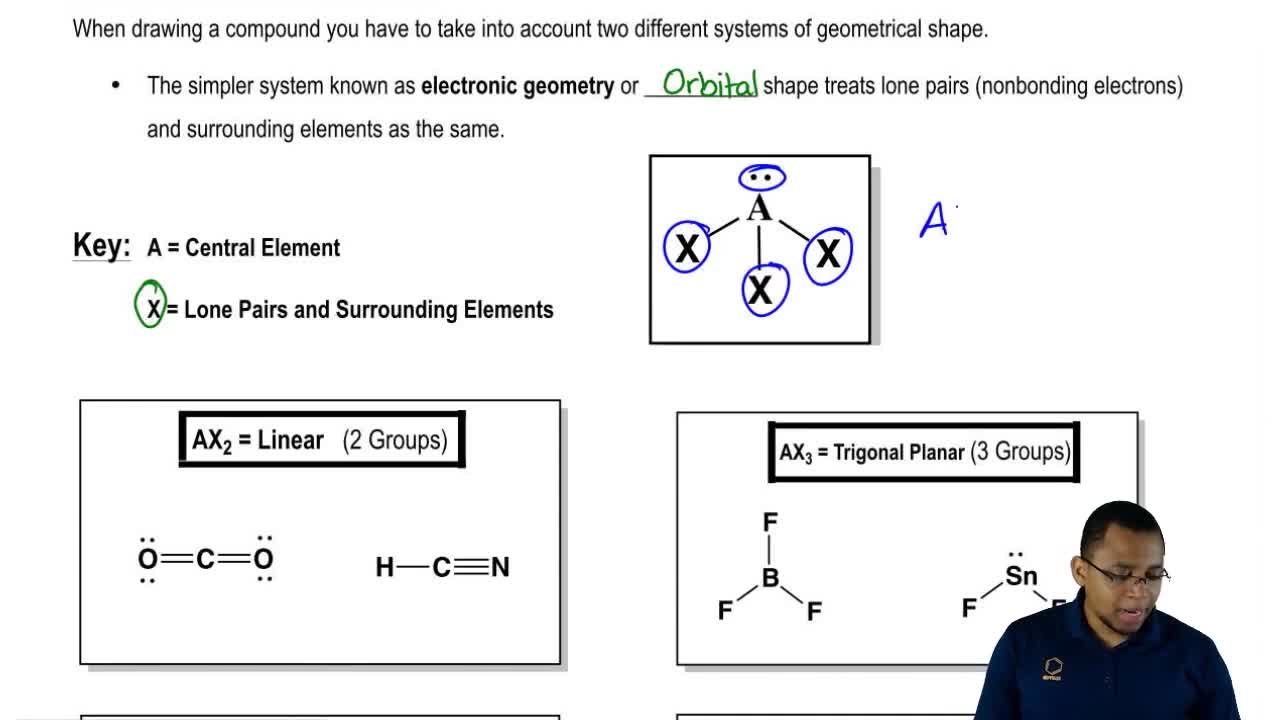
Question 123Predict the molecular geometry and polarity of the CS2. According to VSEPR theory, for a given molecule the central atom has to be chosen and the total number of valence electrons must be counted and from the electron count of the central atom the structure of the molecule is predicted. Question 115What is the molecular shape of HOF as predicted by the VSEPR theory. The VSEPR theory Valence shell electron pair repulsion theory is basically a model used to determine the geometry of a molecule from the number of electron pairs surrounding the central atom of the molecule. Using the VSEPR theory, we can determine the number of bond pairs and lone pairs of electrons that are present on the central atom of the given compound and from there try to predict the geometry of the compound. The best way to start when trying to figure out a molecules geometry is its Lewis structure. 
Hint : We know by trying to apply VSEPR theory Valence shell electron pair repulsion theory in this question.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
